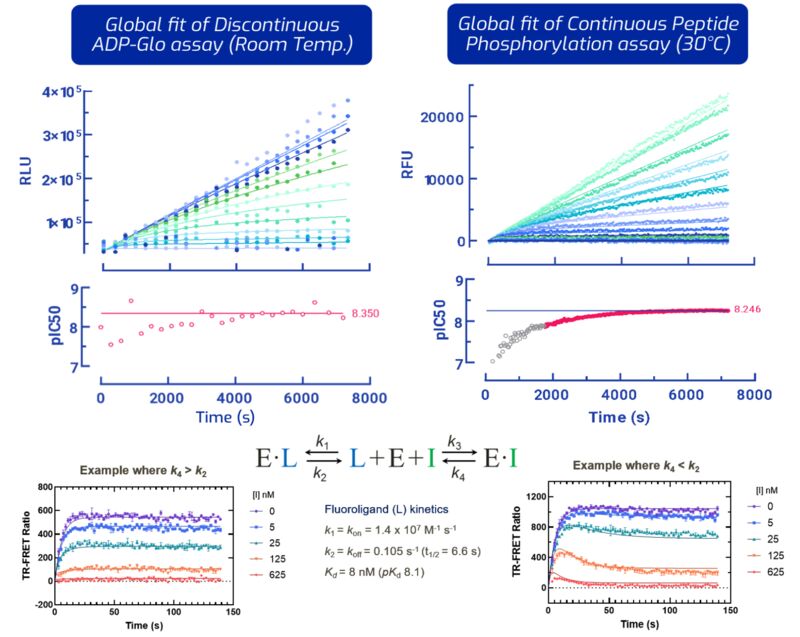
Kinetic analysis plays a central role in drug discovery by characterising the temporal aspects of drug–target interactions. These studies provide quantitative insight into binding and dissociation processes, informing key pharmacological properties such as potency, selectivity, and duration of action.
Predicting drug efficacy: Kinetics can help predict how quickly a drug will bind to its target and how long it will stay bound. This information is critical for determining the drug’s efficacy and optimal dosing regimen.
Optimizing drug design: Kinetic studies can help optimize the design of a drug by providing insights into the relationship between the chemical structure of the drug and its pharmacological properties. For example, knowing the kinetic parameters of a drug can help medicinal chemists design more potent and selective drugs with fewer side effects.
Understanding drug toxicity: Kinetic studies can help identify potential toxicity issues with a drug by revealing how it interacts with other biological molecules and how quickly it is metabolized and eliminated from the body.
Facilitating regulatory approval: Kinetic studies are required by regulatory agencies like the FDA to evaluate the safety and efficacy of new drugs. By providing a detailed understanding of a drug’s pharmacokinetic and pharmacodynamic properties, kinetic studies can help expedite the approval process.
Kinetic analysis represents a fundamental component of modern drug discovery, providing quantitative insight into the dynamic behaviour of drug–target interactions. These data inform the evaluation of pharmacological properties and support the rational design of compounds with improved efficacy and safety profiles. At Arctoris, we use automation to run these experiments as early as possible in the drug discovery journey so that we can identify and optimise the desired properties from the beginning.